
Generation of multipotent foregut stem cells from human pluripotent stem cells. Coordination of morphogenesis and cell-fate specification in development. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Intestinal crypt homeostasis revealed at single-stem-cell level by in vivo live imaging. Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5 + stem cell. Transplantation of expanded fetal intestinal progenitors contributes to colon regeneration after injury. Single Lgr5 stem cells build crypt–villus structures in vitro without a mesenchymal niche. Optimality in the development of intestinal crypts. Hedgehog-responsive mesenchymal clusters direct patterning and emergence of intestinal villi. Villification: how the gut gets its villi. The intestinal stem cell signature identifies colorectal cancer stem cells and predicts disease relapse. Spatial reconstruction of single enterocytes uncovers broad zonation along the intestinal villus axis. Identification of Lgr5-independent spheroid-generating progenitors of the mouse fetal intestinal epithelium. Morphogenesis and compartmentalization of the intestinal crypt. From definitive endoderm to gut-a process of growth and maturation. Parasitic helminths induce fetal-like reversion in the intestinal stem cell niche. YAP/TAZ-dependent reprogramming of colonic epithelium links ECM remodeling to tissue regeneration. Intestinal label-retaining cells are secretory precursors expressing Lgr5. Dll1 + secretory progenitor cells revert to stem cells upon crypt damage. 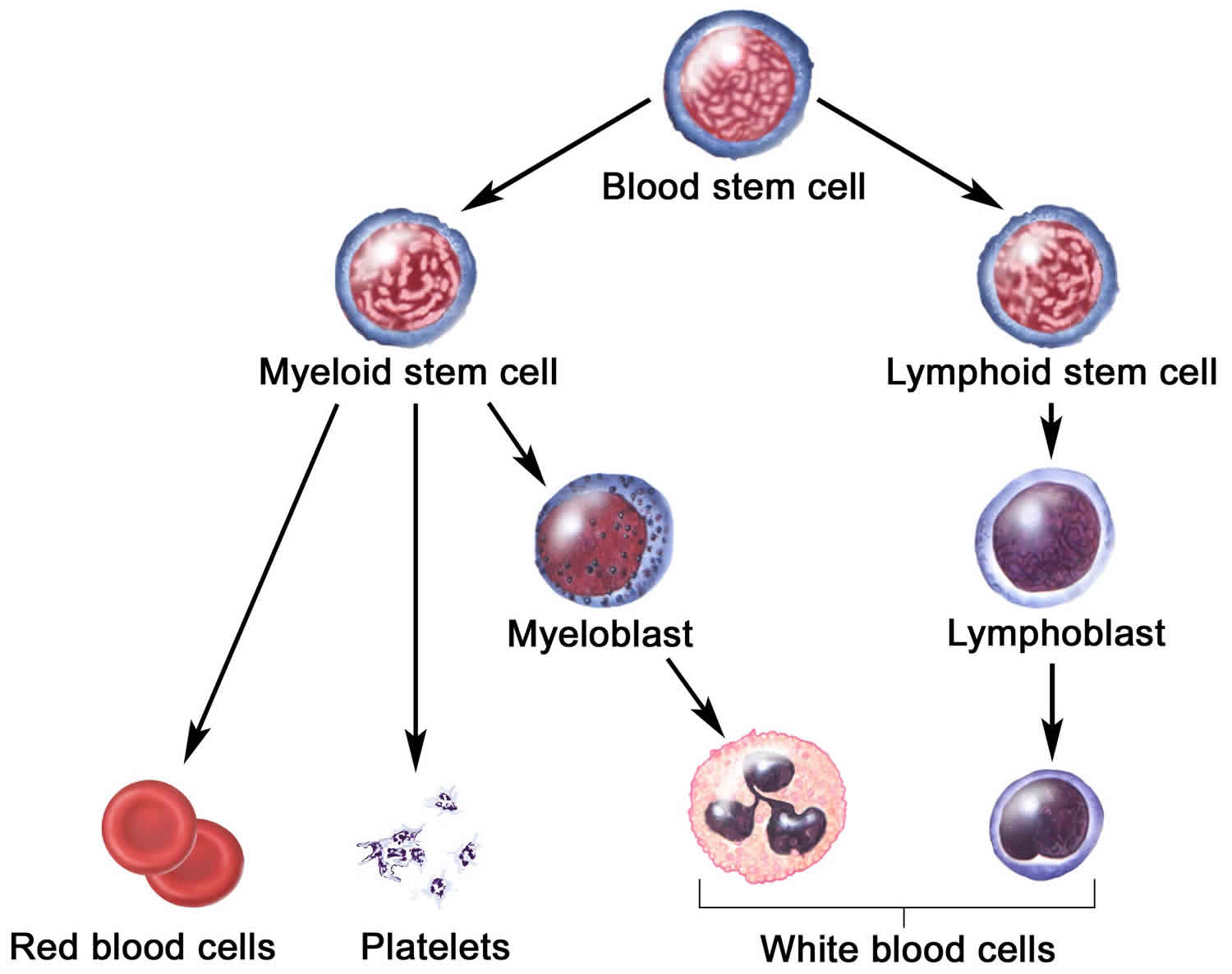
Replacement of lost Lgr5-positive stem cells through plasticity of their enterocyte-lineage daughters. Id2 controls specification of Lgr5 + intestinal stem cell progenitors during gut development. Bending gradients: how the intestinal stem cell gets its home. Identification of stem cells in small intestine and colon by marker gene Lgr5. The intestinal crypt, a prototype stem cell compartment. Moreover, these findings provide a direct link between the observed plasticity and cellular reprogramming of differentiating cells in adult tissues following damage 5, 6, 7, 8, 9, revealing that stem-cell identity is an induced rather than a hardwired property.Ĭlevers, H. Our results demonstrate that large-scale remodelling of the intestinal wall and cell-fate specification are closely linked. This brings epithelial cells from the non-proliferative villus into the proliferative intervillus region, which enables them to contribute to the adult stem-cell niche. 
Using 3D imaging, we find that during fetal development the villus undergoes gross remodelling and fission. Here we show, using unbiased quantitative lineage-tracing approaches, biophysical modelling and intestinal transplantation, that all cells of the mouse intestinal epithelium-irrespective of their location and pattern of LGR5 expression in the fetal gut tube-contribute actively to the adult intestinal stem cell pool. Although fetal LGR5-expressing cells can give rise to adult intestinal stem cells 3, 4, it remains unclear whether this population in the patterned epithelium represents unique intestinal stem-cell precursors. Adult intestinal stem cells are located at the bottom of crypts of Lieberkühn, where they express markers such as LGR5 1, 2 and fuel the constant replenishment of the intestinal epithelium 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
